The Arizona Section of ECS will be hosting a meeting with special guest speaker Professor Robert F. Savinell.
Date: January 26, 2014
Time: Networking and refreshments at 6:15 PM; Seminar begins at 7:00 PM
Place: University of Arizona
Tuscon, AZ 85721
Agave Room, 4th Floor of Student Union Building
Cost: Free to attend; $5 for light refreshments
Speaker: Professor Robert F. Savinell
George S. Dively Professor of Electrochemical Engineering at Case Western Reserve University
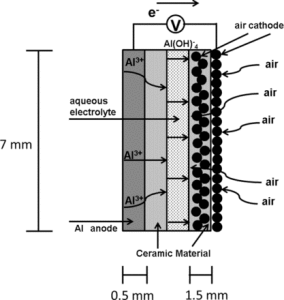
Professor Savinell is recognized as a leading authority on electrochemical energy storage and conversion. His research has been directed at fundamental science and engineering research for electrochemical systems and novel device design, development, and optimization. Dr. Savinell has over 100 publications and seven patents in the electrochemical field. He is a past chair of ECS’s Electrolytic and Electrochemical Engineering Division, a former editor of the Journal of The Electrochemical Society, and a Fellow of ECS.