Sheela Berchmans, CSIR-Central Electrochemical Research Institute chief scientist.
Guest post by: Sheela Berchmans, chief scientist at the CSIR-Central Electrochemical Research Institute
Sheela Berchmans has been an ECS member since 2012 and member of the Organic and Biologic Division and India Section since 2019. Berchmans’ areas of expertise include microbial fuel cells, nanomaterials for sensor applications, bio-assisted synthesis of metal nanoparticles, and electrocatalysis. Read her past work, available now in the ECS Digital Library.
Follow the latest research on electrocatalysis at the 235th ECS Meeting taking place on May 26-30, 2019 in Dallas, TX.
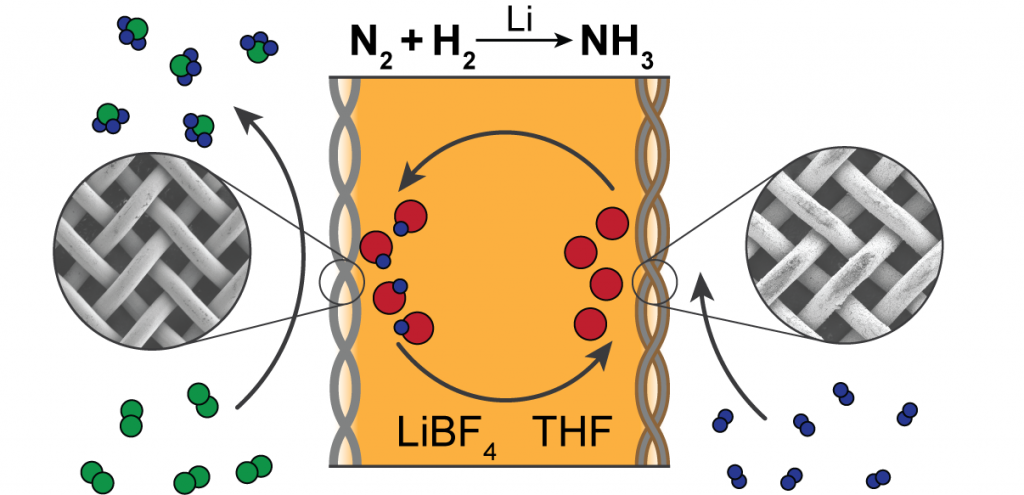
Electrocatalysis assumes a special importance as the applied potential at the electrified interface provides a tunable ∆G to the rate component. ∆G consists of a chemical and a electrochemical component (e-∆G0/RT e-F∆/RT), where the electrochemical component provides a leverage to control the rate of reaction. For simple nonbonding reactions, the rate of the reaction can be expressed as a function of work function of the metal catalyst. However, when bonding reactions are concerned, the adsorption of the reactants at the electrode surface determines the rate of the reaction. For eg, we take into consideration, Hydrogen evolution reaction, (HER) a typical prototype of electrochemical reaction.
The following reaction steps determine the rate of the reaction. The first step involves the proton discharge on the electro catalyst (Volmer reaction) which desorbs either through an electrochemical desorption (Heyrovsky reaction) or chemical desorption from the electrode surface as H2 gas. (2nd and 3rd steps) This reaction is known to be highly exothermic in nature.
(more…)
 Spring symposium on April 22, 2023
Spring symposium on April 22, 2023