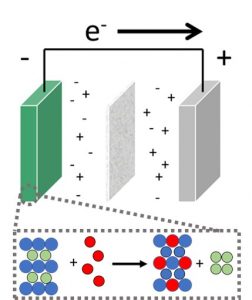
The Electrochemical Society hosted Arumugam Manthiram’s live webinar, “Sustainable Next-generation Battery Chemistries,” on September 20, 2023. Dr. Manthiram took audience questions during a live Question and Answer session at the end of the presentation. He kindly answered in writing questions not answered during the broadcast. His responses follow.
View Dr. Manthiram’s WebinarNOTE: Registration is required to view the webinar.