The Center for Automotive Research (CAR) is conducting the Workforce Skills Assessment of the North American Lithium Ion (Li) Battery Industry in October and November 2023. CAR seeks input from individuals and companies to provide important insights about the skills and training needs for current and future employees.
The Center for Automotive Research (CAR) is conducting the Workforce Skills Assessment of the North American Lithium Ion (Li) Battery Industry in October and November 2023. CAR seeks input from individuals and companies to provide important insights about the skills and training needs for current and future employees.
The assessment’s goal is to gain a better understanding of the evolving employment needs within companies across the battery industry and identify any skills gaps that may be supported by further training and education.
Feedback from knowledgeable individuals is needed. Please click here to access the assessment.* Submissions are accepted until November 3 at 1700h EST.




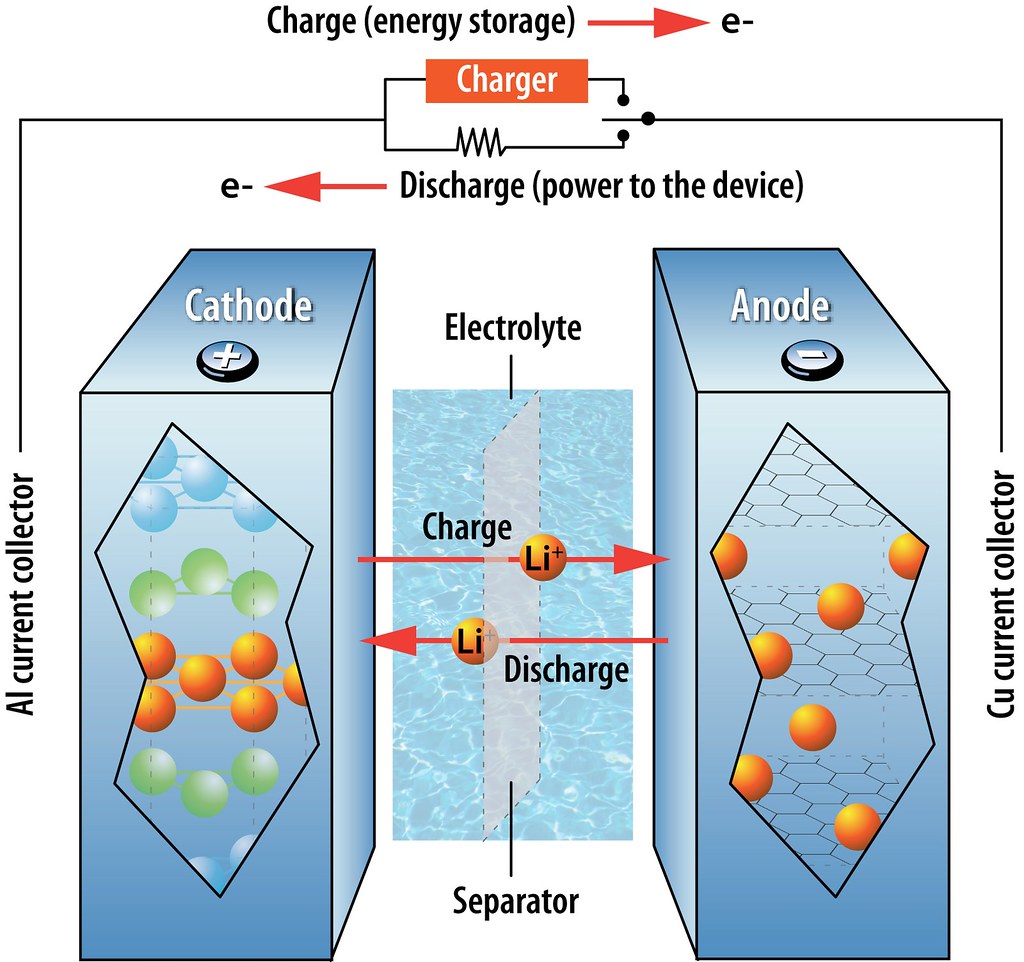
 On June 17, 2020,
On June 17, 2020, 


 The Electrochemical Society honors 2019 Nobel Chemistry Prize laureates, John B. Goodenough, M. Stanley Whittingham, and Akira Yoshino, by the launch of a
The Electrochemical Society honors 2019 Nobel Chemistry Prize laureates, John B. Goodenough, M. Stanley Whittingham, and Akira Yoshino, by the launch of a