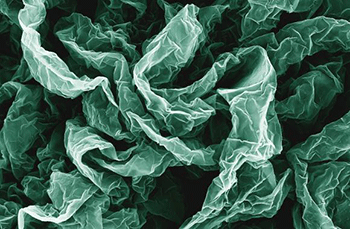
Wrinkles and crumples, introduced by placing graphene on shrinky polymers, can enhance graphene’s properties.
Image: Brown University
By now we’ve heard about the seemingly endless possibilities for the wonder material graphene. The engineers at Brown University are looking to make those possibilities even more appealing through a process that could make the nanomaterial both water repellant and enhance its electrochemical properties.
The research team is looking to improve upon the already impressive graphene by wrinkling and crumpling sheets of the material by placing it on shrink polymers to enhance its properties, potentially leading to new breakthroughs in batteries and fuel cells.
This from Brown University:
This new research builds on previous work done by Robert Hurt and Ian Wong, from Brown’s School of Engineering. The team had previously showed that by introducing wrinkles into graphene, they could make substrates for culturing cells that were more similar to the complex environments in which cells grow in the body. For this latest work, the researchers led by Po-Yen Chen, a Hibbit postdoctoral fellow, wanted to build more complex architectures incorporating both wrinkles and crumples.
Crumpling the graphene makes it superhydrophobic, a property that could be used to develop self-cleaning surfaces. Additionally, the enhanced electrochemical properties could be used in next-generation energy storage and production.
“You don’t need a new material to do it,” said Po-Yen Chen, co-author of the study. “You just need to crumple the graphene.”