“Katsenite” named after McGill researcher who analyzed short-lived material’s chemical structure.
Source: McGill University
Have you heard of mechanochemistry yet? Researchers from McGill University are making a name for themselves in this up-and-coming multidisciplinary field with their discovery of a new material unveiled through unconventional means.
Prof. Tomislav Friščić’s research group in McGill’s Department of Chemistry is now producing chemical reactions through milling, grinding, or shering solid state ingredients. In other words, the team is using brute force to elicit these reactions rather than the typical liquid agents.
The group states that their process is similar to that of a coffee grinder. The advantage to using force over liquids is that it avoids environmentally harmful bulk solvents that are typically used when producing chemical reactions.
These findings were published in the paper “In Situ X-ray diffraction monitoring of a mechanochemical reaction reveals a unique topology metal-organic framework”. It all began in late 2012, where researchers reported that they had been able to observe milling reaction in real time – seeing chemical transformations using highly penetrating X-rays.
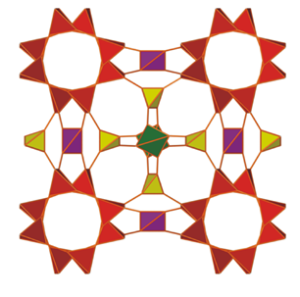
The new study adopts this observed technique to unveil a structurally unusual metal-organic material.
This from McGill University:
The scientists dub the material “katsenite,” after the first author of the article, Athanassios D. Katsenis. Now a postdoctoral fellow at McGill, Katsenis was a visiting student in Friščić’s group when the research was conducted. He analyzed the topology of the material — the arrangement and connections between the structural ‘nodes’) of its crystal structure — and realized that it didn’t correspond to anything previously seen.
This confirms something that scientists have long thought but have not been able to prove: milling creates temporary phases with chemical structure that are not achievable under conventional conditions.
To learn more about the cross-section between chemistry and solid state science, make sure to attend the 227th ECS Meeting this May. Registration for this meeting is now open!
And make sure to check out the research on mechanochemistry in our Digital Library!

