Maria Kelly
Sandia National Laboratories
The Electrochemical Society hosted “Challenges in CO2 Reduction Selectivity Measurements by Hydrodynamic Methods,” a live webinar by Maria Kelly (Sandia National Laboratories), on March 18, 2026. A live Question and Answer session followed.
The author’s written responses to questions not addressed during the broadcast follow.
Replay Webinar
Q&A
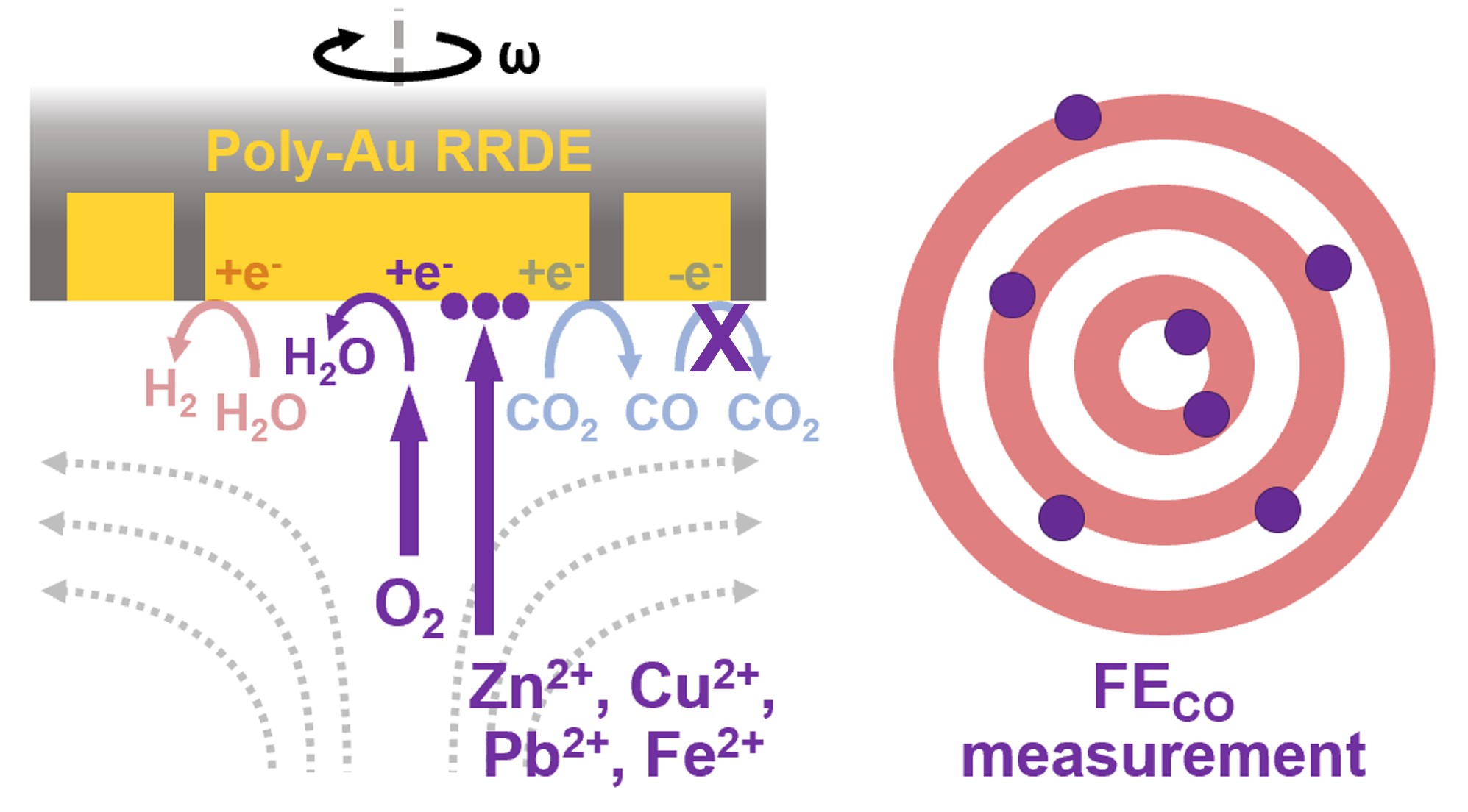
Thirty percent deactivation attributed to the cathodic disk is due to impurities. How does that potentially scale for flow-cell or MEAs?
It’s generally observed across different electrolysis systems that as the surface area of the electrode increases, it becomes less sensitive to contamination effects. Since the metal contaminants in our electrolyte are estimated to be in the ppb to ppt range, this level of contamination is concerning for a low-surface-area rotating disk electrode but not so much for a high-surface-area electrode used in an MEA (microelectrode array). Perhaps over long operational time, you would see effects of contamination in the MEA performance, but I wouldn’t expect it in the initial startup of the system as we see in our RRDE data.
Any estimates on the ppm of the contaminates?
Unfortunately, we didn’t have much luck quantifying the contaminant levels by inductively coupled plasma (ICP). The levels were quite close to our “blank” sample, so we didn’t feel confident reporting those values. According to the manufacturer specification for the trace metal grade Na2CO3 we used for our electrolytes, the contaminants we detected by x-ray photoelectron spectroscopy are at most 0.01 ppm Zn, 0.005 ppm Cu, 0.05 ppm Fe, and 0.01 ppm Pb. Note that these are contamination levels in the salt, so they would be even lower when mixed into a 0.1 M solution. Based on this, I would say the contaminants are in the ppt to ppb range in our electrolyte solution.
How do bubbles at high current densities affect ring measurements?
Gas bubble formation and accumulation at the insulating PEEK spacer between the ring and disk significantly hinders detection of CO at the ring electrode. To get around this problem, we use a method from Marc Koper’s Group to increase the hydrophilicity of the insulating spacer and eliminate bubble attachment (see Vos, J.G. and Koper, M.T.M, J. Electroanal. Chem. 2019, 850, 113363). We simply soak the electrode in a sodium phosphate buffer (pH ≈ 7) containing dopamine hydrochloride to form a polydopamine coating on the electrode surface. The coating is removed from the metal electrode surfaces by electropolishing and leaves only the insulating PEEK modified by the hydrophilic polydopamine.
What about the other product formation during CO2RR?
Several reports have used RRDE to detect CO2RR products other than CO before (see Zhu, X. et al. Electrochim. Acta 2018, 283, 1037–1044) but not quantitatively. I think it is possible though, to attain quantitative selectivity measurements for other products if you can select a ring material that has a very specific/selective mechanism for detecting the product and inactive for all other products. This could include functionalizing the Au ring with a sensing molecule as has been done for pH sensing (see Liu, X., et al., Phys. Chem. Chem. Phys. 2023, 25, 2897–2906). It’s a tricky problem though and would require some careful experiments to prove the chosen sensing method is stable and reliable.
Learn more about upcoming ECS Webinars and review previous webinar recordings.
Interested in presenting in the ECS Webinar Series? Email your presentation title and abstract to education@electrochem.org for consideration.