By: Jeffrey Gardner, University of Maryland, Baltimore County
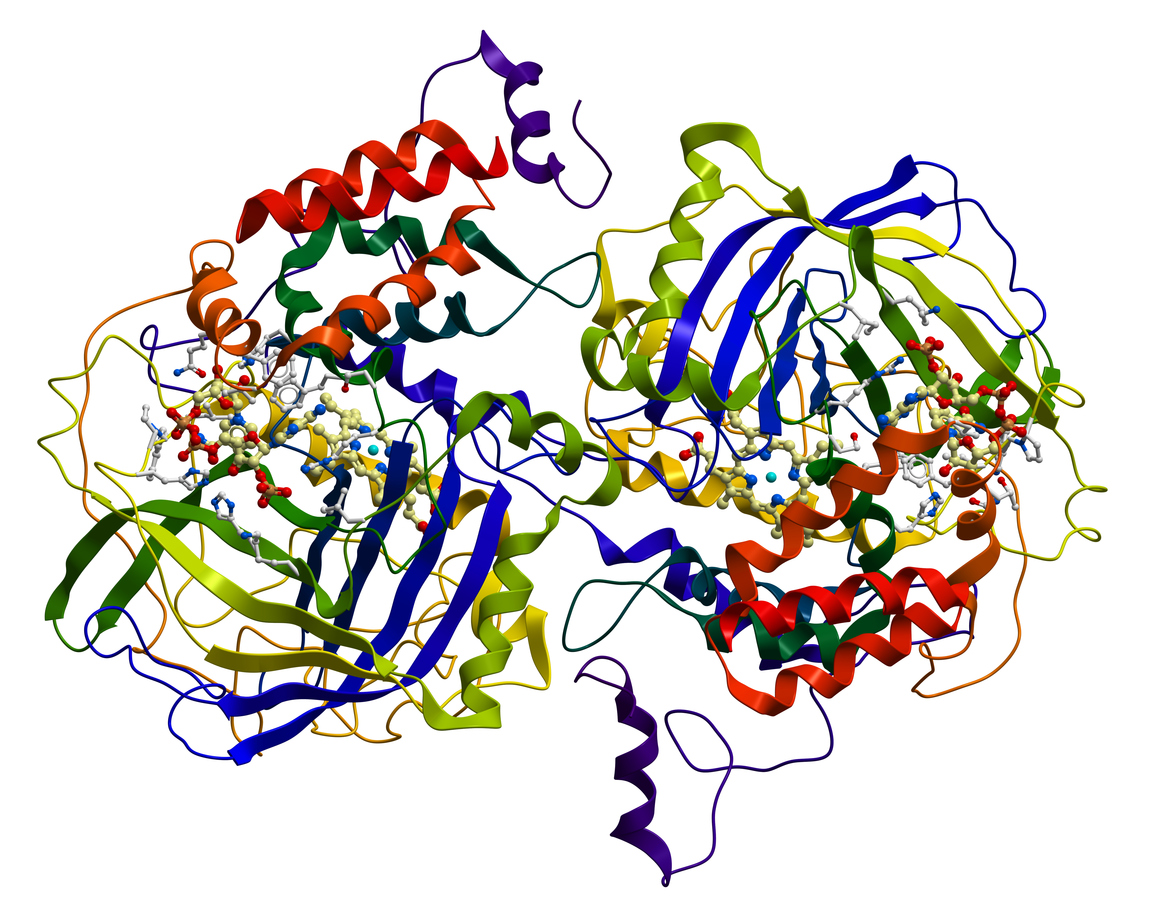 When people hear about prospecting, they might imagine old forty-niners (miners) with pickaxes hunting for gold, or maybe an agent for the San Francisco 49ers (football team) scouting for new talent. In my lab we do another version, called bio-prospecting – searching for useful substances from natural sources. Bio-prospecting has produced many valuable products, including anti-cancer drugs derived from plants and extremely strong silks spun by tropical spiders.
When people hear about prospecting, they might imagine old forty-niners (miners) with pickaxes hunting for gold, or maybe an agent for the San Francisco 49ers (football team) scouting for new talent. In my lab we do another version, called bio-prospecting – searching for useful substances from natural sources. Bio-prospecting has produced many valuable products, including anti-cancer drugs derived from plants and extremely strong silks spun by tropical spiders. ![]()
Our work focuses on enzymes, which are proteins that speed up chemical reactions. We are looking for new and powerful enzymes that can break apart polysaccharides – common molecules that consist of long chains of sugars. Polysaccharides are extremely abundant in the fruits and vegetables that we eat, the cotton clothes we wear and the lumber we use to build houses.
Enzymes that can break down polysaccharides have many uses – for example, in detergents that dissolve stains on clothes. Similar types of enzymes can also be used to release sugars found in plants, which can then be used for manufacturing biodegradeable plastic.
In my lab, we are searching for new enzymes that could improve biotechnology for making renewable fuels and chemicals.


 A team of researchers from Texas A&M University is looking to take the negative impact of excessive levels of carbon dioxide in the atmosphere and turn it into a positive with renewable hydrocarbon fuels.
A team of researchers from Texas A&M University is looking to take the negative impact of excessive levels of carbon dioxide in the atmosphere and turn it into a positive with renewable hydrocarbon fuels.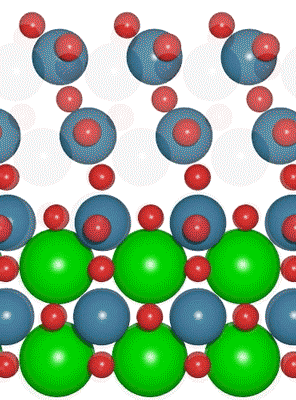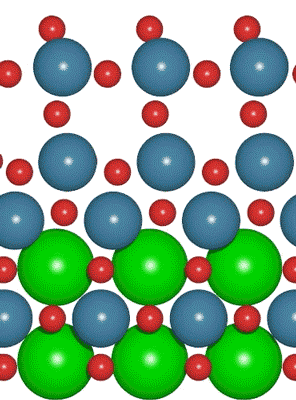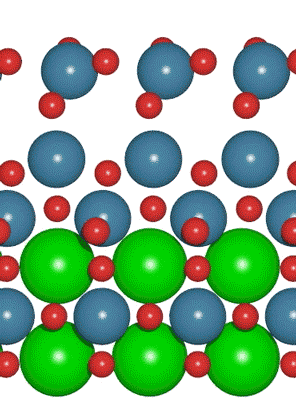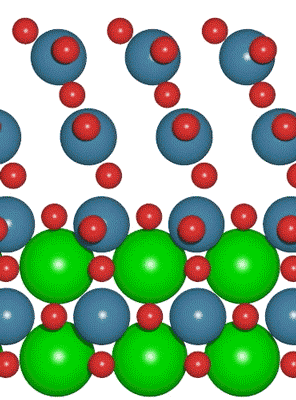 An interdisciplinary team made up of researchers from Stanford University and the U.S. Department of Energy’s SLAC National Accelerator Laboratory recently developed a new catalyst that carries out a solar-powered reaction 100 times faster than ever before.
An interdisciplinary team made up of researchers from Stanford University and the U.S. Department of Energy’s SLAC National Accelerator Laboratory recently developed a new catalyst that carries out a solar-powered reaction 100 times faster than ever before.