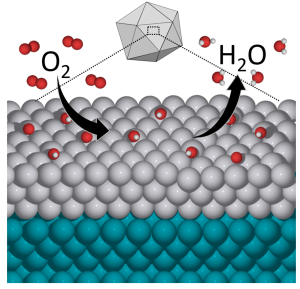
The new catalyst combines platinum and palladium, resulting in high efficiency levels and lower cost.
Image: Mavrikakis group, UW-Madison
In recent years, platinum has been the leading material in the energy industry. However, platinum is both expensive and scarce.
In order to boost alternative energy solutions, researchers have been searching for a substitute for platinum that will allow for cheaper and equally efficient energy technology.
In order to do this, a team from the University of Wisconsin-Madison and Georgia Institute of Technology are focusing on a new catalyst that combines the more expensive platinum with the less expensive palladium.
This from University of Wisconsin-Madison:
This not only reduces the need for platinum but actually proves significantly more catalytically active than pure platinum in the oxygen reduction reaction, a chemical process key to fuel cell energy applications. The palladium-platinum combination also proves more durable, compounding the advantage of getting more reactivity with less material. Just as importantly, the paper offers a way forward for chemical engineers to design still more new catalysts for a broad range of applications by fine-tuning materials on the atomic scale.
“This is speaking to the precise arrangement of atoms on the surface of a nanoparticle,” said Manos Mavrikakis, head researcher in the development. “That can make an enormous difference in how fast the reaction takes place. Theory has been instrumental for about 10 years now to demonstrate the importance of being able to tailor-make specific facets of the same material.”
This new development could make catalysis-driven processes more efficient and less expensive. By moving away from platinum, there are more opportunities for new energy technologies due to economic feasibility.
PS: Don’t forget to register for the ECS Conference on Electrochemical Energy Conversion & Storage with SOFC-XIV! Check out the lead organizers of the conference, who are among some of the top researchers in their respective fields.


