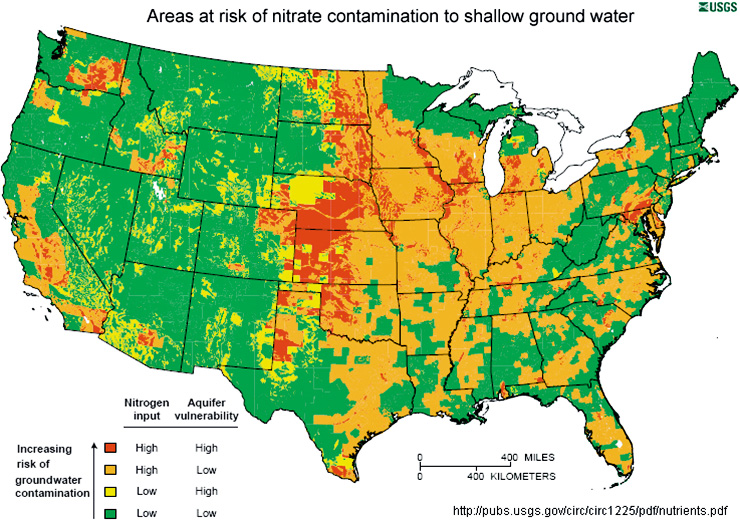
Many areas of the United States are at risk for nitrate and nitrite contamination of drinking water due to overuse of agricultural fertilizers. Click to enlarge.
Image: USGS
Researchers have found a catalyst that can clean toxic nitrates from drinking water by converting them into air and water.
“Nitrates come mainly from agricultural runoff, which affects farming communities all over the world,” says lead study scientist Michael Wong, a chemical engineer at Rice University.
“Nitrates are both an environmental problem and health problem because they’re toxic. There are ion-exchange filters that can remove them from water, but these need to be flushed every few months to reuse them, and when that happens, the flushed water just returns a concentrated dose of nitrates right back into the water supply,” he explains.
Wong’s lab specializes in developing nanoparticle-based catalysts, submicroscopic bits of metal that speed up chemical reactions. In 2013, his group showed that tiny gold spheres dotted with specks of palladium could break apart nitrites, the more toxic chemical cousins of nitrates.


 Match the following figures – Albert Einstein, Thomas Edison, Guglielmo Marconi, Alfred Nobel and Nikola Tesla – with these biographical facts:
Match the following figures – Albert Einstein, Thomas Edison, Guglielmo Marconi, Alfred Nobel and Nikola Tesla – with these biographical facts: Sensors on tape that attach to plants yield new kinds of data about water use for researchers and farmers.
Sensors on tape that attach to plants yield new kinds of data about water use for researchers and farmers.
 This article was originally published in the winter 2017 issue of
This article was originally published in the winter 2017 issue of  In a
In a  Every day about
Every day about  Don’t discount the honorable mention!
Don’t discount the honorable mention! Over 1,840 articles were published in ECS journals in 2017, ranging from battery technology to materials science. Among those articles, “
Over 1,840 articles were published in ECS journals in 2017, ranging from battery technology to materials science. Among those articles, “