 An article by Kenji Amaya, Naoki Yoneya, and Yuki Onishi published in the latest issue of Interface.
An article by Kenji Amaya, Naoki Yoneya, and Yuki Onishi published in the latest issue of Interface.
Protecting structures from corrosion is one of the most important challenges in engineering. Cathodic protection using sacrificial anodes or impressing current from electrodes is applied to many marine structures. Prediction of the corrosion rates of structures and the design of cathodic protection systems have been traditionally based on past experience with a limited number of empirical formulae.
Recently, application of numerical methods such as the boundary element method (BEM) or finite element method (FEM) to corrosion problems has been studied intensively, and these methods have become powerful tools in the study of corrosion problems.
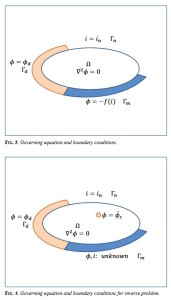
With the progress in numerical simulations, “Inverse Problems” have received a great deal of attention. The “Inverse Problem” is a research methodology pertaining to identifying unknown information from external or indirect observation utilizing a model of the system.