 Each year ECS awards up to five Summer Fellowships to assist students in continuing their graduate work during the summer months in a field of interest to the Society. Congratulations to the five Summer Fellowship recipients for 2014. The Society thanks the Summer Fellowship Committee for their work in reviewing the applications and selecting five excellent recipients. Applications for the 2015 Summer Fellowships are due January 15, 2015.
Each year ECS awards up to five Summer Fellowships to assist students in continuing their graduate work during the summer months in a field of interest to the Society. Congratulations to the five Summer Fellowship recipients for 2014. The Society thanks the Summer Fellowship Committee for their work in reviewing the applications and selecting five excellent recipients. Applications for the 2015 Summer Fellowships are due January 15, 2015.

ECS member Jiaxing Huang used freshman-level chemistry to solve the solubility mystery of graphene oxide films.
Image: Northwestern University
Sometimes science can be extremely complex and commanded by technical expertise. But there are moments when one has to go back to his roots to find a more simple answer for a complex issue. That is what ECS member Jiaxing Huang – along with a team of Northwestern University researchers – has done in order to solve the mystery that surrounds the solubility of graphene oxide films.
For years, one question has puzzled the materials science community – why are graphene oxide (GO) films highly stable in water?
When submerged, GO sheets become negatively charged and repel, which should cause membrane to disintegrate. Though much to the confusion of the scientific community, when GO sheets are submerged they stabilize.
ECS’s job board keeps you up-to-date with the latest career opportunities in electrochemical and solid state science. Check out the latest openings that have been added to the board.
P.S. Employers can post open positions for free!
Director of Publications
The Electrochemical Society – Pennington, New Jersey
Serves as senior staff member responsible for the overall strategic direction of the ECS publications (journals, ECS Transactions, and Interface) and all content in the ECS Digital Library. Assists with the creation and implementation of special projects and initiatives that advance the mission of the organization, which is to provide the greatest possible dissemination of the technical content. Strives to make ECS the top publisher in electrochemical and solid state science, maintaining consistency with ECS mission, goals, and objectives.
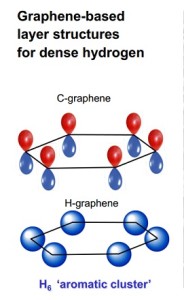
A comparison of the basic ring structure of the carbon compound graphene with that of a similar hydrogen-based structure synthesized by Carnegie scientists.
Credit: Carnegie Science
A new study shows remarkable parallels between hydrogen and graphene under extreme pressures.
The study was conducted by Carnegie’s Ivan Naumov and Russell Hemley, and can be found in the December issue of Accounts of Chemical Research.
Because of hydrogen’s simplicity and abundance, it has long been used as a testing ground for theories of the chemical bond. It is necessary to understand chemical bonding in extreme environments in order to expand our knowledge of a broad range of conditions found in the universe.
It has always been difficult for researchers to observe hydrogen’s behavior under very high pressure, until recently when teams observed the element at pressures of 2-to-3.5 million times the normal atmospheric pressure.
Under this pressure, it transforms into an unexpected structure that consists of layered sheets, rather than close-packed metal – which had been the prediction of scientists many years ago.

Gerischer’s immense contributions continue to leave an indelible mark, not only in electrochemistry, but also in physical chemistry and materials chemistry.
An article by Adam Heller, Dieter Kolb, and Krishnan Rajeshwar in the Fall 2010 issue of Interface.
Heinz Gerischer was born on March 31, 1919 in Wittenberg, Germany. He studied chemistry at the University of Leipzig between 1937 and 1944 with a two-year interruption because of military service. In 1942, he was expelled from the German Army because his mother was born Jewish; he was thus found “undeserving to have a part in the great victories of the German Army.” The war years were difficult for Gerischer and his mother committed suicide on the eve of her 65th birthday, in 1943. His only sister, Ruth (born in 1913), lived underground after escaping from a Gestapo prison and was subsequently killed in an air raid in 1944.
In Leipzig, Gerischer joined the group of Karl Friedrich Bonhoeffer, a member of a distinguished family, members of whom were persecuted and murdered because of opposition to Nazi ideology. Bonhoeffer descended from an illustrious chemical lineage of Wilhelm Ostwald (1853-1932) and Walther Hermann Nernst (1864-1941), and kindled Gerischer’s interest in electrochemistry, supervising his doctoral work on periodic (oscillating) reactions on electrode surfaces, completed in 1946. He followed Bonhoeffer to Berlin where his PhD supervisor had accepted the directorship of the Institute of Physical Chemistry at the Humboldt University, and also became the department head at the Kaiser Wilhelm Institute for Physical Chemistry in Berlin-Dahlem (later the Fritz Haber Institute). Gerischer himself was appointed as an “Assistent.” Many years later, Gerischer would return to this distinguished institution as its director. With the Berlin Blockade and the prevailing economic conditions the post-war research was carried out under extremely difficult conditions.
Request for Proposals
The Electrochemical Society with Toyota North America
Announces the ECS Toyota Young Investigator Fellowship
for Projects in Green Energy Technology
Proposal Submission Deadline: January 31, 2015

ECS, in partnership with the Toyota Research Institute of North America (TRINA), a division of Toyota Motor Engineering & Manufacturing North America, Inc. (TEMA), is requesting proposals from young professors and scholars pursuing innovative electrochemical research in green energy technology.
Global development of industry and technology in the 20th century, increased production of vehicles and the growing population have resulted in massive consumption of fossil fuels. Today, the automotive industry faces three challenges regarding environmental and energy issues: (1) finding a viable alternative energy source as a replacement for oil, (2) reducing CO2 emissions and (3) preventing air pollution. Although the demand for oil alternatives—such as natural gas, electricity and hydrogen—may grow, each alternative energy source has its disadvantages. Currently, oil remains the main source of automotive fuel; however, further research and development of alternative energies may bring change.
 Imagine a world where anyone—from the student in Atlanta to the researcher in Port au Prince—can freely read the scientific papers they need to make a discovery, where scientific breakthroughs in energy conversion, sensors or nanotechnology are unimpeded by fees to access or publish research.
Imagine a world where anyone—from the student in Atlanta to the researcher in Port au Prince—can freely read the scientific papers they need to make a discovery, where scientific breakthroughs in energy conversion, sensors or nanotechnology are unimpeded by fees to access or publish research.
At ECS, that is our vision of the future. We’re working to provide open access to all ECS publications, while maintaining our high standards of peer-review and fast delivery of content.
Please help us make this vision a reality by
making a tax-deductible donation to ECS today.
Your donation fosters the growth of electrochemistry and solid state science and technology by supporting ECS publications and the participation of scientists from around the world at our biannual meetings.
Through travel grants and reduced fees, ECS enables the participation of young scientists and students who otherwise might not be able to attend an ECS meeting. This is particularly important as the work of these scientists, and all ECS members, increasingly holds the keys to solving global challenges in energy, waste and sustainability.
Please help us continue the important work of ECS by donating today.
Thank you again for your incredible work and continued support.
ECS’s job board keeps you up-to-date with the latest career opportunities in electrochemical and solid-state science. Check out the latest openings that have been added to the board.
P.S. Employers can post open positions for free!
Copper Electrodeposition for Via Filling
Osaka Prefecture University – Sakai, Japan
The researcher will be engaged in the development of new electrodeposition process for three dimensional packaging including TSV process; design copper deposition bath containing appropriate additives and fabricate copper filled deep vias on silicon wafer; use analytical techniques such as cyclic voltammetry, chronoamperometry, SEM, XRD, and so on. Presents of research at international conferences and publish in peer-reviewed journals are encouraged upon approval from collaborating companies and institutes.

Madeleine Jacobs, Chief Executive Officer and Executive Director of the American Chemical Society.
Credit: Peter Cutts
Madeline Jacobs has held steadfast to the idea of improving lives through the transforming power of chemistry during her career. Now, after seeing all she set out accomplish during her time as the American Chemical Society’s director and chief executive officer come to fruition, Jacobs is ready to move on to something new.
Jacobs started her career with the American Chemical Society (ACS) as a reporter for Chemical & Engineering News in 1969. Here, she rose in the ranks – becoming the magazine’s editor-in-chief in 1995.
Her work at Chemical & Engineering News prepared her for the role that she would take on in 2003 as ACS’s executive director.
A recently conducted experiment may give us a better understanding of how the Earth possibly began.
Scientists took to the lab with a powerful 500-foot laser to re-create what might have been the original spark of life on Earth.
This from Associated Press:
The researchers zapped clay and a chemical soup with the laser to simulate the energy of a speeding asteroid smashing into the planet. They ended up creating what can be considered crucial pieces of the building blocks of life.



