Hosted by ECS Pacific Northwest Section
A video recording of The Pacific Northwest Hydrogen Workshop webinar is now available for open-access viewing on the ECS YouTube channel.
The Society and ECS Pacific Northwest Section hosted the workshop on July 21, 2022. This timely event covered:
- Roles of hydrogen and hydrogen technologies in decarbonization of our society;
- Status of industrial deployment of hydrogen technologies, (e.g., fuel cells, electrolyzer);
- R&D needs and workforce development for hydrogen technologies



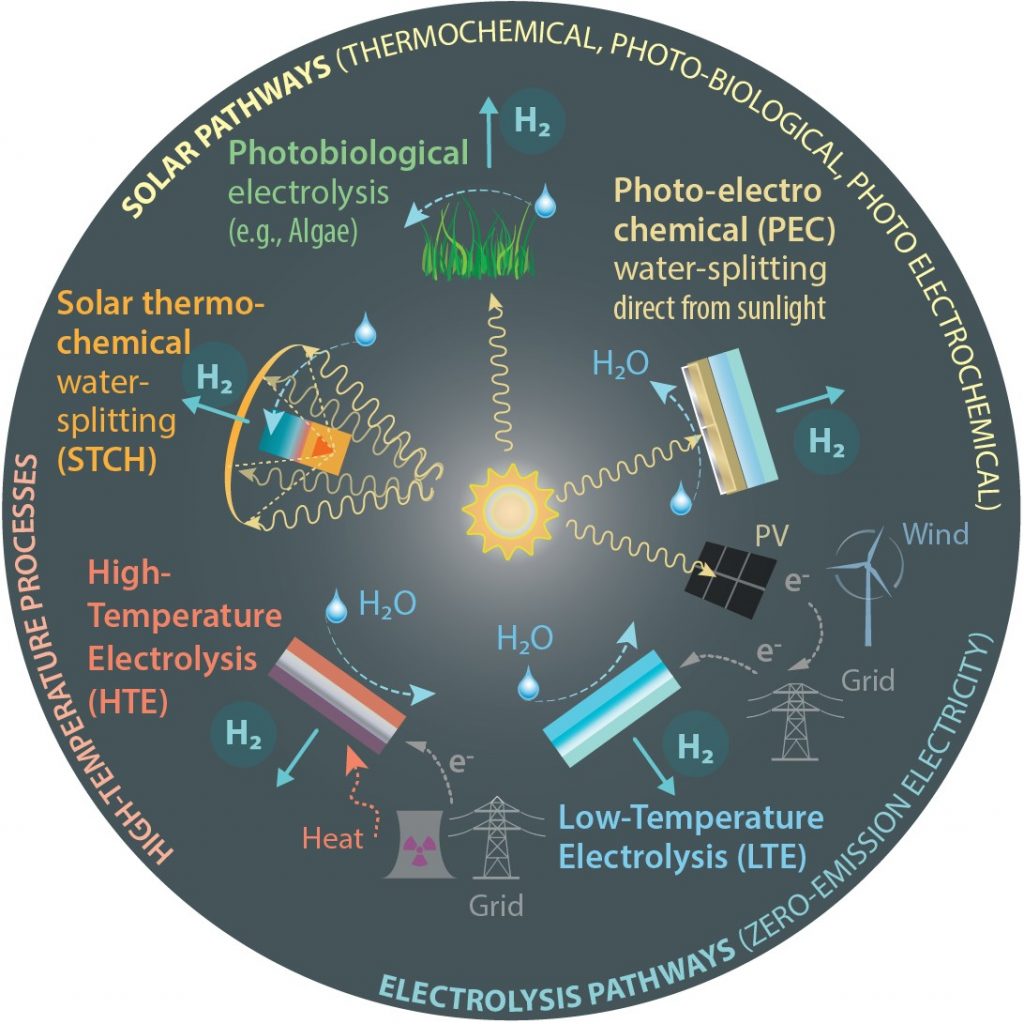
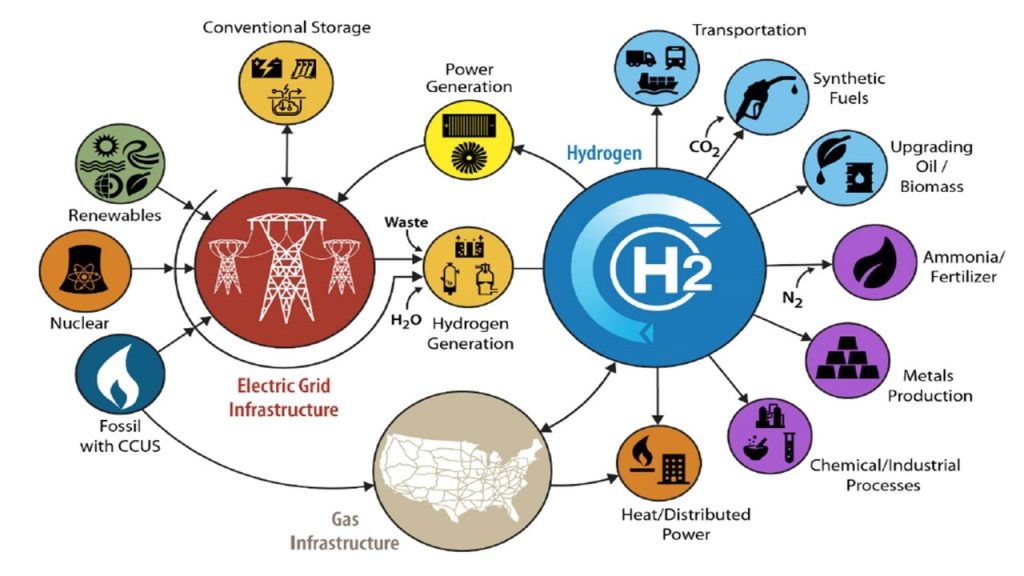
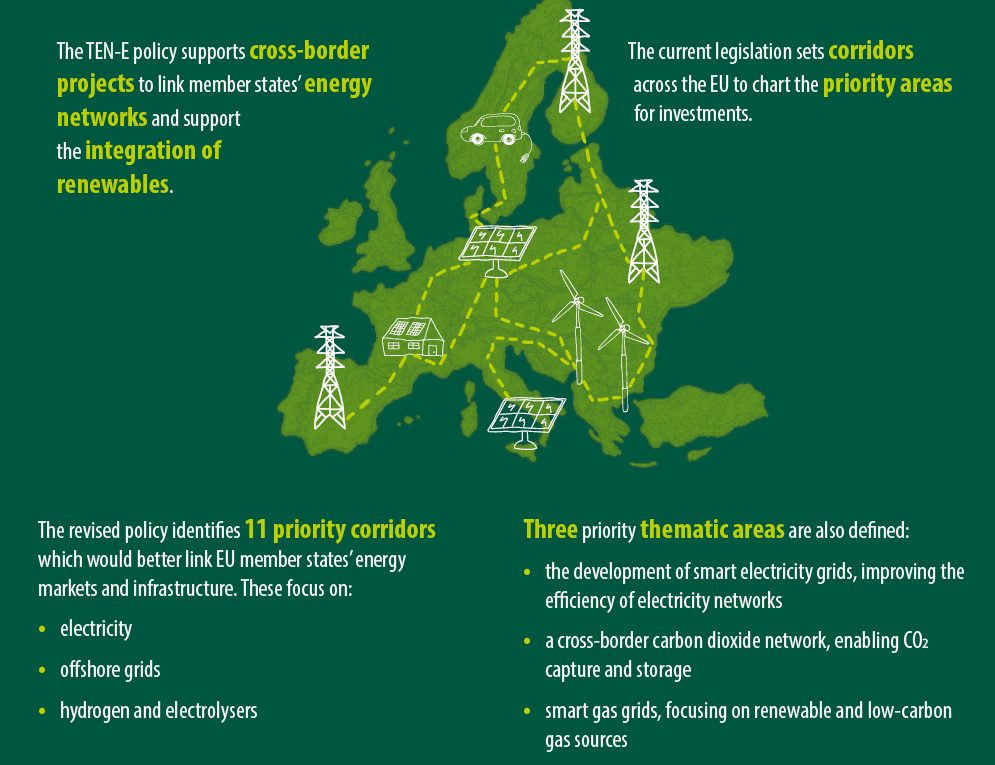




 Researchers at KTH have successfully tested a new material that can be used for cheap and large-scale production of hydrogen – a promising alternative to fossil fuel.
Researchers at KTH have successfully tested a new material that can be used for cheap and large-scale production of hydrogen – a promising alternative to fossil fuel.